|
ClostraBio offers versatile solutions for delivery of bacterial metabolites.
|
PROBIOTIC SUPPLEMENT
CLB-101 is a butyrate-producing probiotic found to be lacking in infants with food allergy THERAPEUTIC ASSET
CLB-004 treats underlying condition in ulcerative colitis (UC): modulating barrier function and immune response |
Non-communicable chronic diseases are associated with impaired barrier function
Due to an altered intestinal microbiome
Due to an altered intestinal microbiome
Disruption of tight junctions & mucus film covering the epithelial layer causes increased permeability of the intestinal epithelium & increased uptake of luminal or food antigens, ultimately leading to chronic inflammation.
Modern lifestyle factors, including diets low in dietary fiber, results in a loss of gut microbial diversity linked to decreased levels of key bacterial metabolites and impaired intestinal barrier function.
Iweala and Nagler, Annual Reviews Immunology 2019; Vijay and Valdes, European Journal of Clinical Nutrition 2022; van der Hee and Wells, Trends in Microbiology 2021
Modern lifestyle factors, including diets low in dietary fiber, results in a loss of gut microbial diversity linked to decreased levels of key bacterial metabolites and impaired intestinal barrier function.
Iweala and Nagler, Annual Reviews Immunology 2019; Vijay and Valdes, European Journal of Clinical Nutrition 2022; van der Hee and Wells, Trends in Microbiology 2021
Hypothesis: directly deliver bacterial metabolites
Utilize butyrate to correct impaired intestinal barrier function
Utilize butyrate to correct impaired intestinal barrier function
|
In preclinical models, butyrate-producing bacteria in the Clostridia class protect against intestinal inflammation
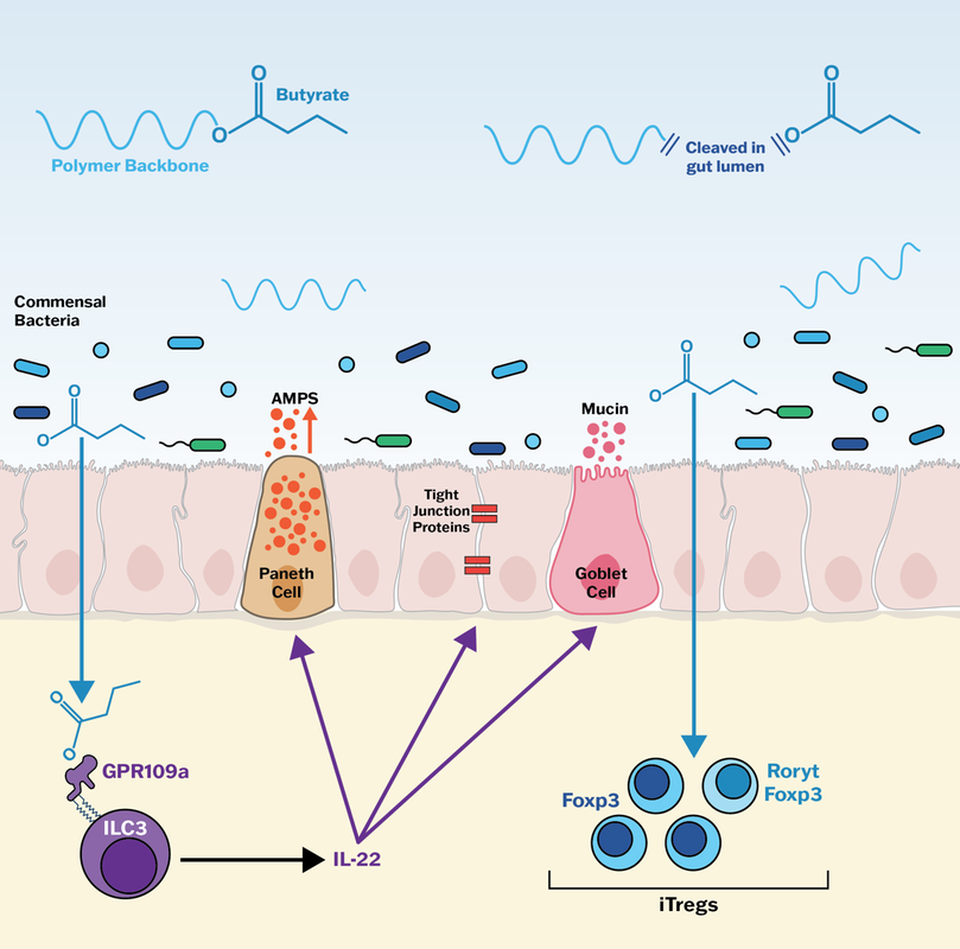
Butyrate induces IL-22, a barrier protective cytokine
Butyrate supports:
|
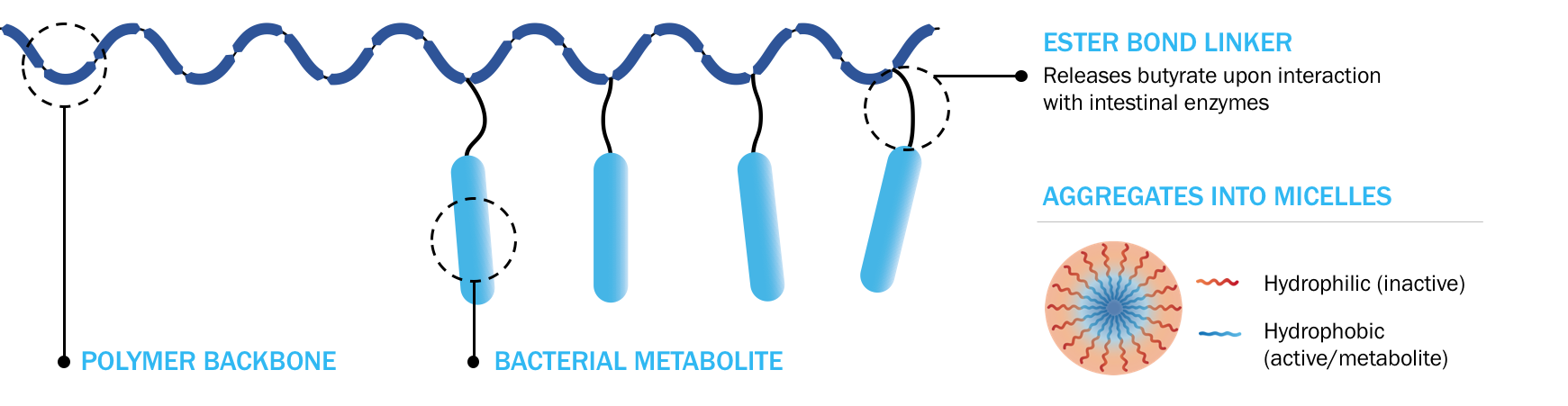
Solution: Proprietary, oral, targeted metabolite delivery platform
Enables butyrate to achieve its maximum therapeutic potential
Enables butyrate to achieve its maximum therapeutic potential
Proposed CLB-004 mechanism
Support innate immune function and stabilize the gut microbiome
Support innate immune function and stabilize the gut microbiome
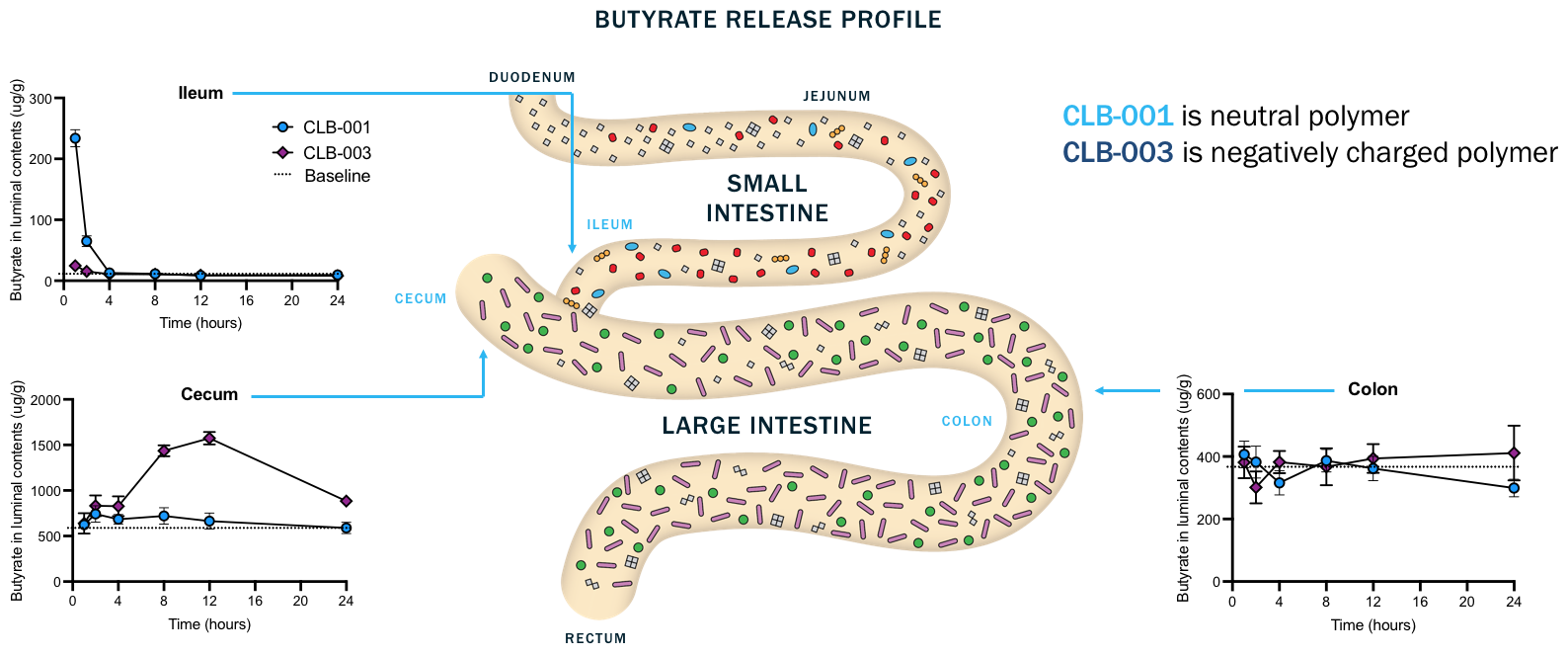
Different polymer constructs enable release in key GI tract locations
Where butyrate-producing bacteria reside
Where butyrate-producing bacteria reside
ClostraBio’s lead CLB-004 candidate is composed of a mixture of CLB-001 and CLB-003.
Note: Vertical axes vary. n= 9/10 mice/timepoint
Note: Vertical axes vary. n= 9/10 mice/timepoint
Treatment for mild-to-moderate UC patients is limited
Butyrate is a safe option but with several delivery constraints
Butyrate is a safe option but with several delivery constraints
*bwhcrohnscolitis.org; Figure from Chris Howerton, PhD. Equity Research Analyst – Biotechnology. Jeffries LLC. Presented at IBD Innovate: Product Development for Crohn’s & Colitis™, Nov 19, 2021.
|
Summary
|
CLB-004 has demonstrated in vivo evidence in multiple species and disease models including ulcerative colitis & food allergy
|
|
CLB-004 has low safety risk: no tox signal in 28-day GLP studies due to use of safe bacterial metabolites and innocuous polymer block components
|
Manufacturing process transferred to CDMO and reproduced at pilot scale
|
|
Positive pre-IND regulatory feedback: standard 505(b)(1) path to IND
|
Phase 1 clinical design includes mild-moderate ulcerative colitis patients to provide early exploratory efficacy evidence
|
|
Both ulcerative colitis and follow-on food allergy indication are focused on patient populations within defensibly large market opportunities and significant unmet needs
|
Plug-n-play platform enables pipeline with multiple small molecule targets
|